Overview
Background
For most of the 20th century, from 1909 until the late 1990s, the premier treatment for symptomatic benign prostatic hypertrophy (BPH) was transurethral resection of the prostate (TURP). TURP was the first successful, minimally invasive surgical procedure of the modern era. To this day, it remains the criterion standard therapy for obstructive prostatic hypertrophy and is both the surgical treatment of choice and the standard of care when other methods fail.
Since the advent of medical therapy for symptomatic prostatic hypertrophy with 5-alpha reductase inhibitors and alpha-adrenergic blockers, the need for immediate surgical intervention in symptomatic prostatic obstruction has been reduced substantially. However, alpha-blockers do not modify prostate growth, and even the use of prostatic growth inhibitors such as finasteride or dutasteride often fails to prevent recurrent urinary symptoms of BPH and retention. In the past, these patients would almost certainly have undergone TURP years earlier.
The criteria for performing TURP surgery are now more stringent than before. In general, TURP surgery is reserved for patients with symptomatic BPH who have acute, recurrent, or chronic urinary retention; in whom medical management and less-invasive prostatic surgical procedures failed; who have prostates of an unusual size or shape (eg, a markedly enlarged median lobe, significant intravesical prostatic encroachment); who have azotemia or renal insufficiency due to prostatic obstruction; or who have the most severe symptoms of prostatism.
Less common uses of TURP include intractable prostatitis or for tissue sampling when standard biopsy techniques cannot be used.
The relative frequency of TURP compared to open prostatectomy in surgical patients varies from country to country. In 1990, the relative frequency rate of TURPs in surgical patients with BPH in the United States was 97%, with similar rates in Denmark and Sweden. The lowest rates of TURP were noted in Japan (70%) and France (69%).
The average age of patients currently undergoing TURP is approximately 69 years, and the average amount of prostate tissue resected is 22 g. Risk factors associated with increased morbidity include prostate glands larger than 45 g, operative time longer than 90 minutes, and acute urinary retention as the presenting symptom. The 5-year risk rate for a reoperation following TURP is approximately 5%. Overall mortality rates following TURP by a skilled surgeon are virtually 0%.
African Americans more typically present for TURP surgery with urinary retention or urinary infections and have a higher incidence of preexisting medical problems compared to the general population. According to Kang et al (2004), reports from the Prostate, Lung, Colorectal, and Ovarian (PLCO) cancer screening trial indicate that Asian and Asian American men have the lowest overall risk of clinical BPH and eventual TURP.
Go to Prostate Cancer, Prostate-Specific Antigen, and Benign Prostatic Hypertrophy for complete information on these topics.
Indications
According to the Agency for Health Care Policy and Research guidelines for the diagnosis and treatment of BPH and the recommendations of the Second International Consultation on Benign Prostatic Hypertrophy, the absolute indications for primary surgical management of BPH are as follows:
Refractory urinary retention
Recurrent urinary tract infections due to prostatic hypertrophy
Recurrent gross hematuria
Renal insufficiency secondary to bladder outlet obstruction
Bladder calculi
Permanently damaged or weakened bladders
Large bladder diverticula that do not empty well secondary to an enlarged prostate
Most men who present for surgical correction of their urinary outlet obstruction are those in whom medical therapy or alternative procedures have failed or are inappropriate for some reason. In general, patients with moderate-to-severe lower urinary tract obstructive symptoms (American Urological Association [AUA] symptom index >8) who have not responded to alpha-adrenergic blockers and/or 5-alpha reductase inhibitors are also candidates for surgical intervention.
A study by Blanchard et al showed that patients in whom alpha-blocker therapy is ineffective or those in whom it has failed tend to have poorer outcomes after TURP than men who proceed directly to a transurethral resection.
This is presumably from preoperative bladder damage and other risk factors that affect voiding rather than the size of the prostate. Operating time and weight of resected tissue have been documented as the same between the 2 groups; therefore, prostatic size alone does not account for the difference in outcomes.
Although persistent, progressive, or bothersome symptoms of urinary obstruction due to prostatic hypertrophy that are refractory to medical therapy constitute the most common indication for TURP, 70% of men undergoing the procedure have multiple indications. Patients with prostates larger than 45 g, who present with acute urinary retention, or who require operating times in excess of 90 minutes, are at increased risk for postoperative complications.
Surgical treatment of BPH is also indicated in cases of renal failure or insufficiency secondary to prostatic obstruction. Catheter drainage is usually recommended in such cases until the renal failure resolves. As many as 10% of men with BPH present with some degree of renal insufficiency.
Contraindications
The only absolute indication for an open prostatectomy over a TURP is the need for an additional open procedure on the bladder that must be performed at the same time as the prostatectomy. Such indications include open surgical resection of a large bladder diverticulum or removal of a bladder stone that cannot be easily fragmented by intracorporeal lithotripsy.
A relative indication for the selection of an open prostate surgery over a TURP is generally based on prostatic volume and the ability of the surgeon to complete the TURP in less than 90 minutes of actual operating time (although < 60 min is considered optimal).
In general, open prostatectomy can be justified in a patient with a prostate of 45 g or larger, but this is totally dependent on the skill and experience of the endoscopic urological surgeon. Most experienced urologists use a prostatic volume of 60-100 g as the upper limit amenable to endoscopic removal, but some highly skilled resectionists are capable of safely treating a 200-g prostate with TURP in less than 90 minutes.
Declining Frequency of TURP
The new availability of reasonable alternative medical and surgical treatment options means that TURP, once one of the most commonly practiced urological procedures, is now performed much less frequently. In 1962, TURP operations accounted for more than 50% of all major surgical procedures performed by urologists in the United States. By 1986, this had declined to 38%.
The 1985 Veterans Administration Normative Aging Study estimated the lifetime probability of surgical intervention for prostatic enlargement at 29%, and the 1986 National Health Survey estimated that 350,000 patients in the Medicare age group had a TURP that year, compared to fewer than 200,000 in that same age group by 1998.
These numbers should be considered within the context that the median age of the typical patient is rising (the number of older men with BPH-related symptoms in the United States is expected to increase from 5 million to 9 million persons by 2025), the size of the average resected prostate gland is increasing, and the typical patient has more comorbidities and is generally less healthy than surgical patients of the past.
A comprehensive review of transurethral prostatectomy in the Medicare beneficiaries by Wasson et al compared a national sample from 1991-1997 to a similar group for the period 1984-1990 found that the more recent group demonstrated a substantial decline in the number of TURP surgeries: 50% for white men and 40% for black men.
Compared to the peak period of TURP use in the 1980s, a higher proportion of the men undergoing the procedure were older in the more recent period, with 53% aged 75 years or older.
Another factor that must be considered when evaluating the general decline in the number of TURP procedures performed is the significant reduction in financial reimbursement to urologists for TURP surgeries in the United States.
Physician reimbursement from Medicare for a TURP has dropped from a high of $2000-$3000 in the past to approximately $650 today, with a 90-day global period that covers all postoperative care by the surgeon for 3 months. In many instances, performing a TURP is simply not profitable for the urologist when office overhead, billing and malpractice costs are considered, especially when complications occur.
Alternative surgical procedures, such as microwave therapy and prostatic laser surgery, are reimbursed at much higher levels, even though they may not be as durable or effective. This creates a strong financial disincentive for urologists to perform TURP procedures, except when no reasonable alternatives exist. A 2002 article by Donnell examined the history of Medicare policies and the effect of changes in Medicare reimbursement on TURP.
In a large Canadian series reported by Borth et al, the number of TURP procedures dropped by 60% between 1988 and 1998, presumably because of medical therapy, despite an increase of 16% in the male population older than 50 years.
While the number of patients presenting with urinary retention was significantly higher in the 1998 group than in the 1988 cohort (55% vs 23%), no significant difference was noted in their average age, medical comorbidities, operative parameters, average size of prostate tissue resected, or complication rates.
Anatomic Considerations
The prostate is divided into 3 zones: peripheral, central, and transition. The peripheral zone is the largest of the zones, encompassing approximately 75% of the total prostate glandular tissue in men without BPH. Most prostate cancers originate in the peripheral zone. It is located posteriorly and extends laterally on either side of the urethra.
The central zone is smaller and extends primarily around the ejaculatory ducts. It differs from the peripheral zone primarily in cytologic details and architecture.
The transition zone is usually the smallest of the 3: it occupies only 5% of the prostate volume in men younger than 30 years. This is the zone thought to be the origin of BPH. The transition zone consists of two separate lobes on either side of the urethra and usually involves a small grouping of ductal tissue near the central portion of the prostatic urethra near the internal sphincter.
As the transition zone expands, it can comprise up to 95% of the prostate volume, compressing the other zones. Intraoperatively, the 2 enlarged lobes of the transition zone can be seen obstructing the prostatic urethra on either side. Thus, the term lateral lobes is often used intraoperatively for this tissue to distinguish it from any hyperplastic periurethral gland tissue.
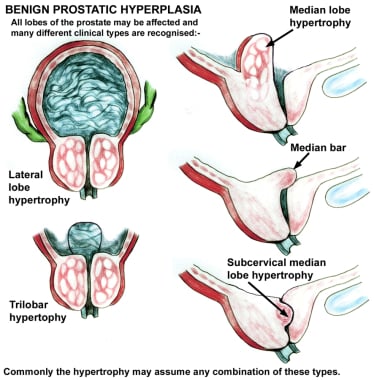
The periurethral glands are less commonly involved with BPH, but when they do become enlarged, they can form what is termed a median lobe, which appears as a teardrop-shaped midline structure at the posterior bladder neck. This can ball-valve into the urethra, creating severe obstructive voiding symptoms. Any significant intravesical extension of prostatic tissue can act as a valve when the detrusor pressure increases and presses this tissue against the bladder neck or across the outlet to the urethra, creating a functional obstruction (see the image below).
Benign prostatic hypertrophy of the lateral and median lobes. Various configurations.
In some earlier jargon, the transition zone and periurethral region were called the central gland or inner gland, and the peripheral and central zones were called the outer gland. This terminology should be avoided both because it is vague and because it creates confusion with the now-standard anatomical label of the central zone.
Prostatic calculi are formed from calcification of the corpora amylacea and precipitation of prostatic secretions. They occur between the transition zone and the compressed peripheral zone; in fact, they can be used as a marker for this border. Prostatic calculi are generally composed of calcium phosphate and are not considered clinically significant. Chemical analysis is unnecessary.
Although prostatic calculi may arise spontaneously, they also may be formed in response to an inflammatory reaction or as a consequence of another pathological process that produces acinar obstruction. Some practitioners believe that calcifications that form in response to bacterial prostatitis may harbor bacteria that periodically flourish, causing recurrent prostatitis. Proponents of this theory advocate TURP to liberalize these calcifications as a treatment for recurrent prostatitis.
If a channel is opened during surgery that allows these calculi to be expressed, they often flow out by themselves if the opening is large enough. They can be milked out by using the end of the cutting loop without current to gently press around the opening where the prostatic stones are seen and can be pushed into the opened prostatic fossa. They can be rinsed into the bladder and evacuated with the rest of the resected prostatic chips.
The prostate is thinnest and most narrow anteriorly (the 12-o’clock position when viewed through a cystoscope). Care should be taken when operating in this area to avoid perforating the prostatic capsule, especially if this portion of the prostate is resected early in the operation. Abundant venous blood vessels are located in the area just anterior to the prostatic capsule, which can cause significant bleeding that cannot be easily controlled if the vessels are damaged during resection.
The external sphincter muscle tends to be slightly tilted, with the most proximal portion located anteriorly, opposite the verumontanum. The external sphincter can be identified cystoscopically by its wrinkling and constricting action as the resectoscope is withdrawn. Upon reinsertion, the superficial mucosa in front of the telescope tends to bunch up. This is because the external sphincter muscle is imbedded within the urogenital diaphragm, which is relatively fixed in position, while the prostate has some limited mobility.
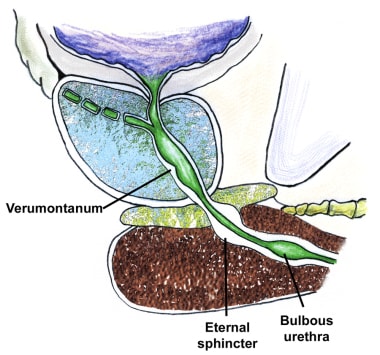
The verumontanum is the single most important anatomical landmark in TURP (see the image below). It is a midline structure located on the floor of the distal prostatic urethra just proximal to the external sphincter muscle. It appears as a small, rounded hump that is best seen when withdrawing the telescope through the prostate while visualizing the prostatic floor at the 6-o’clock position.
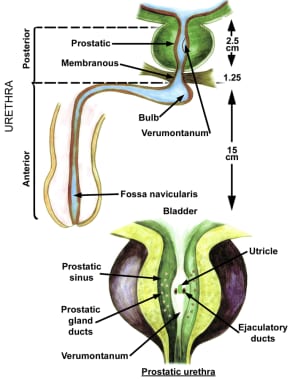
Basic anatomy of the prostate, sagittal section.
The orifices to the ejaculatory ducts emerge in the verumontanum (see the first image below). Its importance lies in its position immediately proximal to the external sphincter muscle (see the second image below), which allows it to be used as the distal landmark for prostate resection. The precise distance between the verumontanum and the external sphincter demonstrates some slight individual variation and should be verified visually before starting the resection and periodically during the surgery.
Anatomy of the prostate and bladder, posterior view.
Anatomy of the prostate and urethra.
The proximity of the ureteral orifices to the cephalad margin of the hypertrophied prostate varies, particularly in patients with an enlarged median lobe. This distance should be frequently assessed throughout surgery.
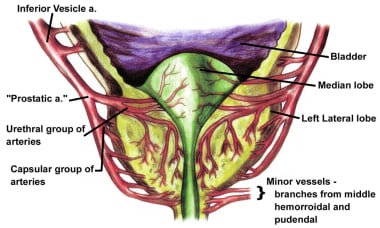
The vascular anatomy of the prostate was accurately described in detail by Rubin Flocks in 1937.
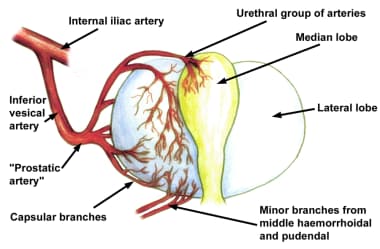
The blood supply of the prostate comes primarily from branches of the inferior vesical artery, which is a branch of the internal iliac artery (see the image below).
Blood supply to the prostate.
When the inferior vesical artery reaches the prostate just at the vesicoprostatic border, it branches into 2 groups of arteries (see the image below). One penetrating group passes directly into the prostate toward the interior of the bladder neck. Upon reaching the prostatic interior near the urethra, most of these branches turn distally and parallel the prostatic urethra, while others supply the median lobe.
Blood supply to the prostate. Note the two main branches: urethral and capsular.
Vessels that parallel the prostatic urethra supply most of the blood to the hypertrophied lateral lobes. The second large group of arteries follows the exterior of the prostatic capsule posterolaterally, periodically giving rise to perforating vessels, and supplies the area around the verumontanum.
See Prostate Anatomy and Male Urethra Anatomy for more information.
Development of Benign Prostatic Hyperplasia
Mechanisms of disease
The prostate has been described as the organ of the body most likely to be involved with disease of some sort in men older than 60 years. This statement characterizes any histological evidence of BPH as a disease, which is certainly debatable, but there is no argument that BPH is an extremely common clinical entity.
As the hyperplastic process increases the volume of the prostate, the urethral lumen is compressed, causing outlet obstruction. An enlarged median lobe may cause relatively more severe symptoms than lateral lobe hyperplasia of similar magnitude because it can act as a valve at which increased bladder pressure may actually cause further obstruction. Intravesical extension of the lateral lobes may act in a similar fashion.
It has been known for many years, however, that prostate size alone is not a reliable or accurate predictor of the presence or degree of urinary outlet obstruction. The failure of several purely obstructive therapies, such as prostatic balloon dilatation, and the obvious success of alpha-adrenergic blockers eventually led to the description of BPH as having both a dynamic (neurogenic) and a mechanical (obstructive) component.
Thus, at the same time as the occurrence of mechanical obstruction, a dynamic component involving the stromal prostatic tissue and bladder is present, which is often more significant in causing urinary symptoms than simple mechanical obstruction from an enlarged prostate. The precise interaction of these two mechanisms, mechanical and dynamic, is not well understood.
When a bladder is trying to empty through a blocked outlet from an obstructing prostate gland, the intravesical pressure required to open the bladder neck is increased. The bladder is initially able to produce a higher transitory voiding pressure when required, but loses muscle tone over time.
Isolated muscle bundles hypertrophy in response to the need for a higher intravesical pressure to overcome the increased resistance to voiding, and bladder trabeculation often follows. The spaces between these hypertrophied bundles tend to become thinner, with less functional muscle. Eventually, this can progress to the point at which the bladder becomes almost nonfunctional.
Bladder trabeculation is usually graded on a scale of I-IV. When seen on cystoscopy images, it is a relative indicator of the degree and duration of any bladder outlet obstruction (eg, BPH), although any detrusor hyperactivity problem can possibly produce bladder trabeculations, even without an identifiable obstruction. Initial symptomatic changes include increased bladder instability and irritability, which can eventually progress to muscular decompensation with permanent loss of detrusor contractile ability.
Evidence indicates that obstruction causes partial denervation of bladder smooth muscle, which results in further bladder irritability and involuntary detrusor contractions. Fortunately, most of these hyperactive symptoms resolve over time with removal of the prostatic obstruction or with a response to appropriate medications. The detrusor becomes less able to maintain a constant voiding pressure over time, which leads to early termination of voiding, intermittency of the urinary stream, and higher residual urine volume, accompanied by loss of bladder compliance.
Overall bladder mass increases because of detrusor muscle hypertrophy, but collagen deposition is also increased, which eventually contributes to decompensation, urinary retention, and permanent loss of detrusor contractile ability.
Proposed causes
BPH is thought to be caused by aging and by long-term testosterone and dihydrotestosterone (DHT) production, although their precise roles are not completely clear.
Histopathologic evidence of BPH is present in approximately 8% of men in their fourth decade and in 90% of men by their ninth decade. Loss of testosterone early in life prevents the development of BPH. The similarities in presentation, pathological examination findings, and symptoms of BPH among identical twins suggest a hereditary influence.
Once BPH has developed, it tends to progress. Cross-sectional studies based on cadaver autopsies or consecutive patients seen in urology clinics suggest that the growth rate decreases with age. In patients aged 31-50 years, the prostate doubling time averages 4.5 years. In men aged 51-70 years, the prostatic doubling time is approximately 10 years, while in men older than 70 years, the doubling time increases to more than 100 years. Note that these findings may only reflect a selection bias in the sample group.
A 5-year longitudinal study by Rhodes and colleagues of 631 community men aged 40-79 years from Olmsted County, Minnesota demonstrated an average annual prostate growth rate of 1.6%. This remained essentially constant regardless of age, although men with larger prostates tended to have higher growth rates.
The average prostate weighs approximately 20 g by the third decade and remains relatively constant in size and weight unless BPH develops. The typical patient with BPH has a prostate that averages 33 g. Only 4% of the male population ever develops prostates of 100 g or larger. (The largest recorded prostatectomy specimen weighed 820 g. This prostate was removed by open suprapubic prostatectomy. Unfortunately, the patient ultimately died of hemorrhage.)
Symptoms of BPH tend to progress slowly over time in most individuals, with an average annual increase of 0.14-0.44 points per year in the AUA symptom index for men aged 60 years and older. Once BPH has begun, the prostate grows an average of 0.6 mL in volume annually, with a mean decrease in average urinary peak flow rate of 0.2 mL per second each year. Men older than 70 years and those with a baseline peak flow rate less than 10 mL/s tend to have a more rapid and dramatic decline in their peak flow rates over time.
DHT has an affinity for prostate cell androgen receptors that is 5 times greater than that of testosterone. The levels of 5-alpha reductase are increased in the stromal tissue of men with BPH compared to controls. This and other data indicate that DHT is much more important in the development of prostatic hypertrophy than testosterone is.
The success of 5-alpha reductase blockers, such as finasteride and dutasteride, in reducing prostatic size and relieving symptoms seems to confirm this, although it does not explain the relative lack of symptom relief in those with smaller prostate glands treated with these agents.
Clinical manifestations and medical treatment
Classic symptoms of BPH include a slow, intermittent, or weak urinary stream; the sensation of incomplete bladder emptying; double voiding (the need to void within a few seconds or minutes of urinating); postvoid dribbling; urinary frequency; and nocturia. Patients may also present with acute or chronic urinary retention, urinary tract infections, gross hematuria, renal insufficiency, bladder pain, a palpable abdominal mass, or overflow incontinence.
Upon physical examination, the bladder may be palpable during the abdominal examination and the prostate may be enlarged during the digital rectal examination. Symptoms are not necessarily proportional to the size of the prostate on digital rectal examination or transrectal ultrasound findings.
Alpha-adrenergic receptors are present and functional in the stromal smooth muscle of the prostate and especially at the bladder neck. Many studies have documented the success of various alpha-adrenergic blockers in relieving symptoms of BPH. Evidence from the Medical Therapy of Prostate Symptoms Trial indicates that combination therapy with both an alpha-blocker and a 5-alpha reductase inhibitor can delay the progression of symptoms and is more effective over time than either medication alone for reducing symptom scores and improving peak urinary flow rates.