Practice Essentials
Urethral strictures can result from inflammatory, ischemic, or traumatic processes. These processes lead to scar tissue formation; scar tissue contracts and reduces the caliber of the urethral lumen, causing resistance to the antegrade flow of urine.
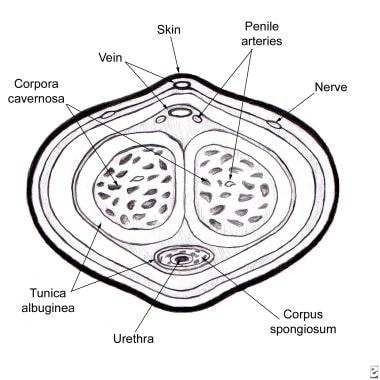
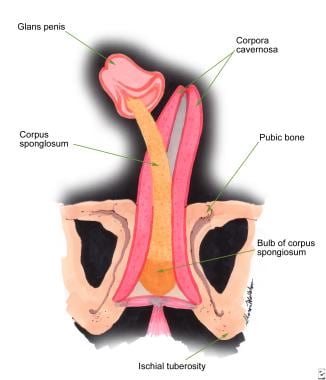
The term urethral stricture generally refers to the anterior urethra and is secondary to scarring in the spongy erectile tissue of the corpus spongiosum. A posterior urethral stricture is due to a fibrotic process that narrows the bladder neck and usually results from a distraction injury secondary to trauma or surgery, such as radical prostatectomy.
The focus of this article is anterior urethral stricture disease.
Urethral strictures arise from various causes and can result in a range of manifestations, from an asymptomatic presentation to severe discomfort secondary to urinary retention. Retrograde urethrogram (RUG) is the main diagnostic method to find anterior urethra stricture and help find the length of the stricture.
Establishing effective drainage of the urinary bladder can be challenging, and a thorough understanding of urethral anatomy and urologic technology is essential. See the images below.
Urethral strictures. Cross-sectional diagram of the penis.
Urethral strictures. Schematic of penile anatomy.
Many techniques are available for the treatment of urethral stricture disease. Based on the literature, no technique can be applied successfully to every situation. Each technique has advantages and disadvantages.
The role of tissue engineering and stem cells in urethral reconstruction
Tissue engineering incorporates the disciplines of cell transplantation, materials science, and engineering with the objective of creating functional replacement tissue. El Kassaby et al published a randomized comparative study of buccal mucosal and acellular bladder matrix grafts. An off-the-shelf matrix derived from the bladder was used. This biomaterial was obtained from donors and prepared via a multistep process, resulting in the removal of all cellular components. The tissue matrix that remains consists of collagen, elastin, growth factors, and macromolecules. Predicated on biocompatibility and the ability to recruit urethral tissue growth in several experimental and clinical studies, this matrix was used.
With a mean follow-up period of 25 months in patients with a healthy urethral bed, the success rates for the acellular bladder matrix were similar to those using buccal mucosa. In patients who had undergone two or more prior urethral surgeries with significant spongiofibrosis, the success rate significantly deteriorated for the acellular matrix relative to buccal mucosa. This study demonstrates promise for the use of acellular matrices as a viable option for urethral repair in patients with a healthy urethral bed, no fibrosis of the corpora spongiosis, and good urethral mucosa.